Laboratory products
Enhanced Seal Performance for Freeze-Drying Applications
Jun 05 2012
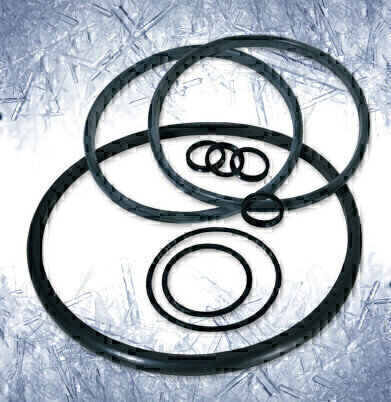
Freeze-drying processes can involve cycling temperatures in the range of - 20°C to +130°C, together with solvents and/or water under medium/high pressures. This combination of aggressive working conditions is conducive to the development of thermal stresses in the seals utilised for the freezer operation. These thermal stresses can also result in associated mechanical stresses leading to premature sealing failures due to cracking and blistering of the seal materials. Dupont™ Kalrez® 6230 perfluoroelastomer (FFKM) parts have been developed to meet such challenging sealing requirements, offering excellent steam cycling resistance up to 260°C.
In freeze-drying applications, the Kalrez® 6230 parts can also be used in a tri-clamp seal. This provides an extended performance in the presence of oxidising chemicals over competitive materials, while operating at high pressures and cycling temperatures between -20°C and +121°C.
Freeze-drying was actively developed for preserving serums being sent to Europe for the medical treatment of the war wounded during WWII. Without refrigeration, many serum supplies were spoiling before reaching the intended recipients. The commercial technique of freeze-drying enabled serum to be rendered chemically stable and so remain viable without having to be refrigerated. The process was also applied to penicillin and bone, and lyophilisation soon became recognised as an established technique for the preservation of biologicals. The process was also applied to penicillin and bone, and lyophilisation soon became recognised as an established technique for the preservation of biologicals. Freeze-drying is now used as a preservation processing technique for a wide variety of food and pharmaceutical products, including vaccines, diagnostic kits, viral or bacterial cultures, tissue analysis, and the production of synthetic skins.
Kalrez® 6230 parts are available in the UK from Dichtomatik Ltd, an authorised UK distributor for Dupont™ Kalrez® products. They are compliant with the United States Food and Drug Administration’s regulations and with the United States Pharmacopeia Class V1 (USP Class V1). They reduce extractables from sealing materials to trace levels, as well as offering a wide range of enhanced performance and endurance capabilities.
Digital Edition
Lab Asia Dec 2025
December 2025
Chromatography Articles- Cutting-edge sample preparation tools help laboratories to stay ahead of the curveMass Spectrometry & Spectroscopy Articles- Unlocking the complexity of metabolomics: Pushi...
View all digital editions
Events
Jan 21 2026 Tokyo, Japan
Jan 28 2026 Tokyo, Japan
Jan 29 2026 New Delhi, India
Feb 07 2026 Boston, MA, USA
Asia Pharma Expo/Asia Lab Expo
Feb 12 2026 Dhaka, Bangladesh